It is generally accepted that for ophthalmic and parenteral administration, isotonic solutions are better tolerated by the patient than those at the extremes of hypo- and hypertonicity to maintain homeostasis with the body’s intracellular fluids.
• Isotonic solution: a solution having the same osmotic pressure as a specific body fluid.
• Hypotonic solution: a solution of lower osmotic pressure than that of body fluids.
• Hypertonic solution: a solution of higher osmotic pressure than that of body fluids.
The calculations involved in preparing isotonic solutions may be made in terms of data relating to the colligative properties of solutions –
• vapor pressure lowering
• freezing point depression
• boiling point elevation
• osmotic pressure
Theoretically, any one of these properties may be used as a basis for determining tonicity. Practically and most conveniently, a comparison of freezing points is used for this purpose. It is generally accepted that - 0.52°C is the freezing point of both blood serum and lacrimal fluid.
Example- 1: Molecular weight of Boric acid is 61.8 thus if 61.8 g in 1000 g of water should produce a freezing point of -1.86°C, calculate the quantity of boric acid is required for a isotonic solution.
1.86/1.52 = 61.8/X
X = 17.3 g
So, 17.3 g of boric acid in 1000 g of water (1.73 %) should make a solution isotonic with lacrimal fluid.
Calculations Formula for Preparation of Isotonic Solution
Example- 2: How much NaCl should be used in preparing 100 ml of 1% w/v solution of atropine sulfate, which is to be made isotonic with lacrimal fluids?
• M.Wt of NaCl = 58.5, i = 1.8
• M.Wt of atropine sulfate = 695, i = 2.6
X = 0.12 g of sodium chloride represented by 1 g of atropine sulfate
Example-3: How many grams of sodium chloride should be used in compounding the following prescription
Rx Pilocarpine nitrate 0.3 g
Sodium chloride q.s.
Purified water 30 ml
Make isoton. Sol.
Sig. for the eye
Sodium Chloride equivalent for Pilocarpine nitrate = (59/271) = 0.22
Step-1. 0.22 X 0.3 = 0.066 g of NaCl represented by the pilocarpine nitrate
Step-2. 0.9 g NaCl for 100 ml water to be isotonic
X g for 30 ml
X = 0.9 X 30 / 100 = 0.27 g
Step-3. 0.27 - 0.066 = 0.204 g of sodium chloride to be used.


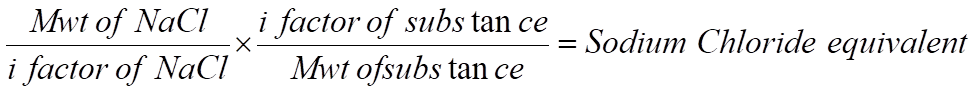
إرسال تعليق